Haseeb Malik, Julie Pham, Nina Reese, Kayla Hogan, Neil Gerstenmaier, Leonard Chen A baby with his brown eyes, a baby with her curly hair. Every prospective parent has hopes for his or her children, long before they are even born. Is it any surprise that parents hope their child could have every advantage in life? Intelligence, charisma, an outgoing personality. These are all traits people hope their children will possess. However, many parents’ hopes are even more basic than what the child will look like or what their personality will be; their only wish is for their child to be born healthy and happy. When this hope is shaken, and the health of a child is called into question before that child ever takes a breath, a parent is faced with some of the hardest decisions of his or her life: either cling to hope or prepare for the worst case scenario. Even then, they may learn their hopes are false, that the life they envisioned for their child is not possible and they must reevaluate how they prioritize the things they once took for granted. What if there was a way to prevent this heartache? A way to flip back the switch and restore these parent’s hopes? What if you could select specific traits that your child would possess? This idea has fascinated society for generations; from novels such as Aldous Huxley’s A Brave New World to movies such as GATTACA, our society has questioned time and again what would happen if we had the power to control our genes, and to some degree our fates. While this has been an interesting hypothetical, it has been pushed into the forefront of the scientific world with the advent of a study in China, led by Junjiu Huang, in which a group used a technique known as CRISPR/CAS9 to edit the genes of human embryos [1]. Although Huang’s study has been discontinued because of issues with using CRISPR, this is the first time in history that CRISPR technology has been used to edit human embryos, a landmark in the journey towards gene editing. When did something that seemed to belong solidly in science fiction become a tangible reality?
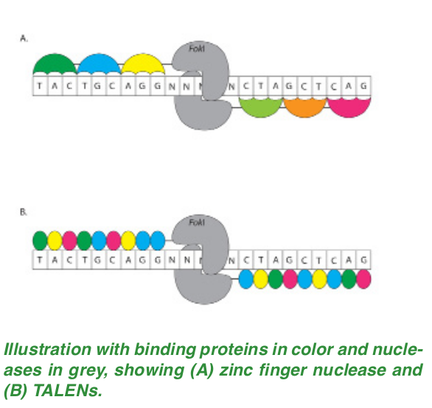
Both the medical and ethical implications for the possible applications of CRISPR/Cas9 have put Dr. Doudna’s work in the spotlight not only in the scientific realm but also in the mainstream media. Since these revolutionary findings, Dr. Doudna has been noted among Time Magazine’s 100 most influential people in the world, and has been invited to events teeming with Hollywood stars. While she has achieved celebrity-status, Dr. Doudna does not seem to be cashing in her retirement early. She recently received $1.5 million from Paul Allen to continue her research on modifying the CRISPR/Cas9 system to attack RNA rather than DNA. All eyes will be on Dr. Doudna as she strives to contribute another medical breakthrough for the fight against genetic diseases. Although Dr. Doudna was the first to start researching CRISPR, she was not granted the patent, leading to a prolonged legal battle on who actually owns CRISPR. Or Is She... Jennifer Doudna and her team used the CRISPR-Cas9 system to target particular genes of their choosing in 2012, filing a patent in March 2013. By the time she had filed the patent, this system was being used in human cells by another group, leading to the potential for use in gene therapies. CRISPR gained popularity among multiple companies, one of which was led by Feng Zhang, a researcher at the Broad Institute and the Massachusetts Institute of Technology. The United States Patent and Trademark Office (USPTO) currently has a first come, first serve system: the first person to file the patent will be granted the patent, regardless of whether they were the first to invent it or not. However, this was only put in place after both parties had filed a patent, making an older rule which stipulates that the first to conceive an idea or put the idea to work will be granted the patent. Zhang filed a patent under a special expedited process and he received the patent first, giving his research center commercial control over its development [3]. Doudna’s original patent had yet to be reviewed. In 2015, Doudna’s team requested the USPTO to determine who developed CRISPR first. When a patent is contested, the process resembles a court case, in which evidence from both labs is reviewed to determine who the rightful patent holder is. Usually, patent disputes are settled through sharing rights to an invention, but this patent dispute does not seem as compromising as other disputes. Cases like these could take years, costing millions of dollars [4]. Even if a decision is reached, an appeal can be made, drawing out the process. Greg Aharonian, the director of the Center for Global Patent Control, can “see many hundreds of thousands of dollars being spent” [5]. There have been previous cases of patent holders placing a fee for taking a license out on patented technology, even among academic researchers. However, the impact that this patent dispute has on those utilizing CRISPR has not yet been determined [6]. The confusion of who owns the rights to the technology may be potentially hindering commercial efforts, delaying products and treatments. Others claim the patent fight is taking the attention away from the most important aspect: the science. The emphasis of the patent is on rights over the technology use and potential Nobel Prize awards, rather than looking at how new methods and applications can be developed with all of this time and money. Already, the number of publications on CRISPR is predicted to surpass 1,100 [5]. Due to CRISPR’s capabilities and potential, the stakes are high with this patent dispute, with only one winner for the rights of the CRISPR system. With the growing attention this technology is receiving, it is easy to forget that CRISPR is not the first gene-editing tool to date. B.C.: Before CRISPR Nucleases have two major components: the first component is the binding protein that recognizes a specific region of the DNA and the second component is the nuclease, which “snips” the DNA and breaks it apart [7]. At this point, a scientist may be able to incorporate a new strand of DNA into the genome. CRISPR uses an enzyme called Cas9, which is a gene-editing nuclease, an enzyme capable of cleaving DNA into its two component strands. Although it now seems to be the nuclease of choice for gene-editing, researchers were initially looking at other options. The first two gene-editing nucleases being explored prior to the discovery of CRISPR are called zinc finger nucleases and TALENs [8]. Zinc Finger Nucleases consist of Zinc Finger Proteins, which make up the binding region of the nuclease; each protein binds to three specific nucleotides. If a Zinc Finger protein fails to bind correctly to its target nucleotide triplets, then the nuclease will be unable to snip the DNA. In this way, the DNA strand is like a barcode, and the Zinc Finger proteins are the scanner which can only react if it finds one barcode. TALENs relies on a similar idea. In TALEN, the binding component of the nuclease is composed of Tal effector proteins, which each bind to one specific nucleotide. This makes it easier to construct TALENs over Zinc Finger nucleases because each tal-effector protein is paired with a specific nucleotide. Zinc Finger nucleases have been challenging to construct at an affordable price while maintaining both high activity and low cytotoxicity [9]. Although TALENs has a low cytotoxicity risk, it has high mutation rates; in other words, it is prone to making mistakes when cutting and adding a DNA sequence to the genome.3 Furthermore, both Zinc Fingers and TALENs require a new nuclease for each location of interest in the genome, making the protocol tedious and inefficient. The process also requires an optimization step to optimize binding to the region of interest, which is time-consuming. CRISPR has the potential to outperform TALEN and Zinc Fingers in both accuracy and speed, gaining the attention of scientists worldwide. 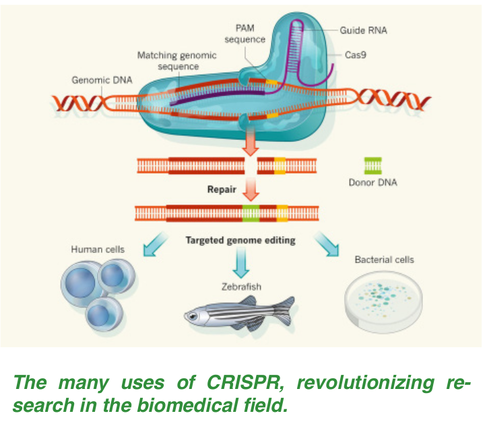 The Mechanism Behind All the Magic The CRISPR-Cas9 enzyme complex, at its core, allows for specificity in cutting DNA sequences and opens up the genome for insertion of desired DNA sequences. It is found naturally in the bacterial immune system as a defense against retroviruses, phages, and viral genome vectors. The basic mechanism involves the CRISPR system using Cas9, the primary endonuclease, as “scissors,” which are guided to the desired site through the use of guide RNA (gRNA) [10]. gRNAs are short RNA transcripts that have a binding site to Cas9 and a complementary binding site for the DNA being targeted. As long as the appropriate gRNA can be created, the Cas9 enzyme can find and target the DNA. Creating the most effective gRNA involves knowing the sequence of the target region on the genome in order to build a unique sequence complementary to the target. The Cas9 complex can then successfully bind to the target DNA and the magic can begin. Using the complementary base pair binding granted by the gRNA, the Cas9 activates and cuts that sequence of DNA [11]. Once the cut has been made, the cell will try to repair this gap using DNA polymerase, but this procedure is error-prone and leads to many mutations. Instead of relying on this natural process, researchers can provide a template for the cell to use to create a new sequence, effectively editing the DNA. This is the main motivation behind using CRISPR: to be able to edit sequences of DNA and potentially remove and repair deleterious alleles that cause disease. Additionally, the wonder of the CRISPR complex is that the gRNA can be modified while the Cas9 does not need to be modified to function with a new gRNA, so they can be used on more sequences of the genome, and multiple CRISPR complexes can be active simultaneously, increasing the efficiency of gene editing. While Cas9 is being touted as the endonuclease that can do it all, researchers believe there is an alternative option in Cpf1 [12]. Cpf1 is a smaller endonuclease than Cas9, making it more effective at entering and working inside cells. Additionally, Cpf1 gives researchers better control over the insertion of the DNA. This method, spearheaded by Feng Zhang of the Broad Institute in Cambridge, Massachusetts, is promising, but understanding whether it is truly applicable is still in the early stages.  The Promises (and Perils of CRISPR-Based Technology Not since the advent of the polymerase chain reaction (PCR) has the field of genomics been so stunningly influenced by a technology as has been recently with CRISPR-mediated gene editing. The emerging applications and theoretical future of this class of techniques, founded upon select methodological patents merely from the last three years alone, are poised to bring about a wealth of industries valued in the billions of dollars. Broadly speaking, this unfolding science will empower research in three fields: medicine, biotechnology, and fundamental biology research [13]. Perhaps the most immediate impacts and benefits of CRISPR technology on human society will come from the field of medicine. Increasingly, medicinal research, both in understanding diseases and improving drug efficacy, relies on our capacity for manipulation on the nanoscale of genetics. Every aspect of biology is encoded in the genome which instructs a given cell to synthesize the biomolecules necessary for its particular role in an organism. The tremendous diversity of life on the planet flows from differences between species’ genetic instructions. Moreover, the ever-challenged survival of a particular genome through reproduction forces an organism to adapt. In an era of little threat from the tusks and teeth of antiquity, our primary enemies are the ubiquitous microbe and the genes involved in heritable diseases and predispositions as well as aging. To meaningfully address these afflictions will require reliable and precise molecular tools, and CRISPR-mediated gene editing promises exactly this. Using CRISPR, researchers can do more to probe the relationship between genes and disease. Because of CRISPR’s unprecedentedly high target-specificity and in vivo (within a living system) efficacy, the current state of gene therapy stands to benefit immensely from this technology [13]. For example, a particular drug for cancer may be ineffective in 40% of the population of lab mice while markedly effective in the rest. One method for understanding this incompatibility would be to alter a single or set of genes of a cell with CRISPR and study its effect on drug action. This way, when certain gene alterations inhibit drug activity, they can be categorized and accounted for in future drug design. In drug research more generally, CRISPR can be used to save time and money by introducing genes that ensure, for example, that a lab mouse will develop a specific type of cancer for therapeutic trials [14]. Ostensibly, CRISPR can even be used to modify embryonic DNA to prevent developing mice, or even humans, from inheriting gene varieties known to be linked to certain health complications. Progress here, however, is likely to be slow considering the ethical conversations and yet-to-be-drafted guidelines necessary for such practice. Although CRISPR is essentially just a way to cut and paste genes, innovative thinkers in the field of bioengineering have proposed using it to create so-called “gene drives.” Generally, a given gene or segment of genes gets passed on by 50/50 chance from either one’s mother or father. Still, some genes can manipulate cell division so as to ensure their own replication. This is referred to as biased-inheritance. A proposed gene drive involves linking select genes of chance with genes of certainty, thereby ensuring the continued replication of any sequence chosen by the scientist [14]. Such a tool could, for example, instigate population-wide fertility loss among mosquitos or simply render the organism immune to the plasmodium parasite responsible for malaria in humans [14]. A CRISPR-powered gene drive would make this existant method vastly more effective as well as economical. Similarly, CRISPR can help increase viral resistance of bacterial strains used in the food industry, such as Streptococcus thermophilus [15]. Like utilizing a hill to roll a large snowball rather than building one by hand, CRISPR allows scientists to utilize exponential reproduction in the wild to spread the tailored genes. Gene drives thus offer the ability to reinvent whole populations of organisms, whether mosquitoes, cash crops, bacteria, or even viruses for human benefit. Such alterations, too, are diverse in their manner; from making an organism more hearty, less infectious, to more industrious in its synthesis or breakdown of biomolecules. CRISPR might just make such feats an everyday feature of the future. In basic biological science, CRISPR can help researchers answer fundamental questions about how genes affect phenotypes (the physical display of a gene or set of genes, such as eye color or color blindness) as well as elucidate the ancestry of certain genes in the tree of life. For example, if a spider carries the exact set of genes found in a fly which, in the latter, are known to be responsible for determining the pattern of wing-vein formation, a reasonable question to ask is, “Why do spiders possess this set of genes?” CRISPR-mediated excision (removal) of this gene set from a spider might lead to physical deformity or instead, exhibit no impact. If the absence of genes were to prove inconsequential, it could be postulated that such genes are vestigial, or leftover, from an evolutionary ancestor of the spider, or perhaps from the last common ancestor of both the spider and the fly [14]. Similar approaches can be applied to a wide range of fundamental questions in biological science. Evolutionary biology and taxonomy (the arranging of species in the tree of life) have come a long way without CRISPR, yet with more questions ahead, the technology will help us better understand the relationships and ancient history of life on Earth. As CRISPR-mediated gene technologies begin to make their way into the bioengineer’s toolbox, their applications and permutations will no doubt expand to unforetold areas of research and society. Though this technology promises pervasive benefits, its power calls for wisdom and forethought. The twenty-first century will be defined by the creativity and extent to which we use the tools of science to create a better future for the human species and the planet. In this way, CRISPR will contribute not just to the sciences, but to the very history of our species.
0 Comments
Leave a Reply. |
Author
Articles written by members of the group on topics they find interesting, exciting, revolutionary. Archives
February 2018
Categories
All
|




 RSS Feed
RSS Feed
